Why as an Optometrist you need to know about Dupixent
Dupixent is a first-in-class medication that was approved for the treatment of moderate-to-severe atopic dermatitis in 2017, and its indications have since been extended to include conditions such as asthma and chronic rhinosinusitis with nasal polyposis.
Dupilumab is a monoclonal antibody that functions as a dual inhibitor of interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling pathways, and it is administered by subcutaneous injection every two weeks. However, its use has been associated with ocular side effects, including photosensitivity, blepharitis, and other ocular surface complications.
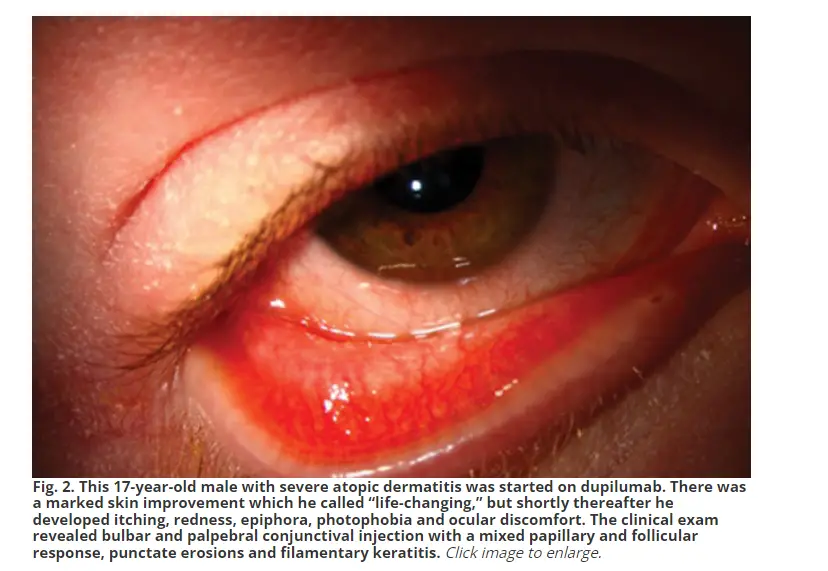
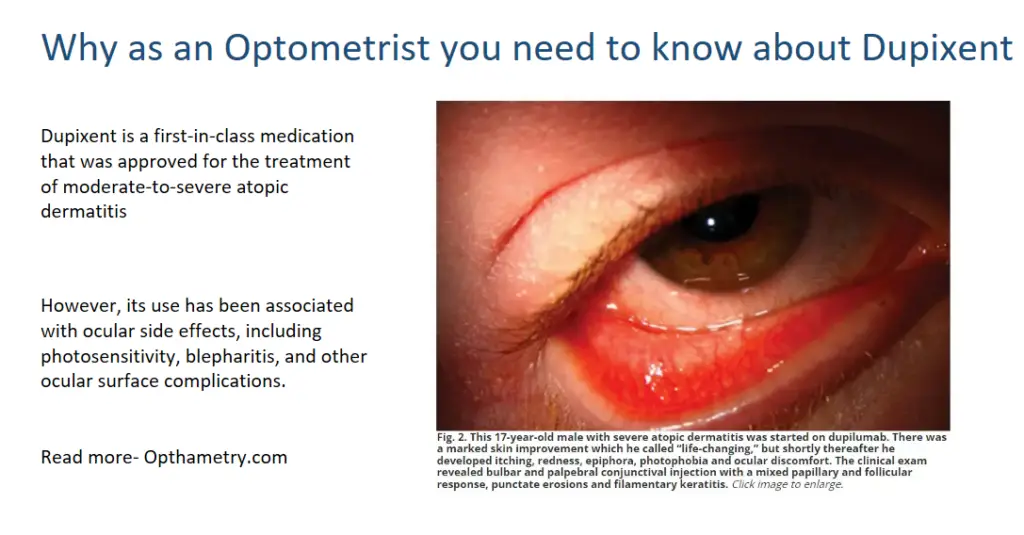
Photosensitivity is one of the ocular side effects associated with Dupixent use. Patients receiving Dupixent treatment have reported symptoms such as chronic redness, dryness, tearing, and itching of both eyes. These symptoms are often accompanied by bilateral conjunctival injection, epiphora, and blepharitis. In some cases, patients have experienced significant improvements in their skin condition, but have also developed ocular discomfort, photophobia, and other ocular symptoms.
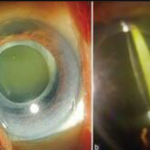
Ocular examination of patients using Dupixent has revealed ocular surface and periocular effects such as conjunctival hyperemia, follicular conjunctivitis, limbal nodules, filamentary keratitis, dry eye, and Demodex infestation. In some cases, more severe complications such as conjunctival cicatricial changes, madarosis, punctal stenosis, cicatricial ectropion, and limbal stem cell deficiency have been observed.
Blepharitis
Blepharitis is another significant ocular complication associated with the use of Dupixent. It has been observed that patients on Dupixent treatment may develop meibomian gland dysfunction, thickened and mildly keratinized lid margins, and inflammatory changes such as papillary and follicular response. The ocular complications associated with Dupixent pose a management challenge for eye care providers.
Treatment of these complications involves careful consideration of the ongoing use of Dupixent and the need to manage the ocular surface disease effectively. Recommendations for management include initiating treatment with topical ocular lubricants and antihistamine-mast cell stabilizers for mild cases, whereas moderate-to-severe cases may require the use of topical steroid drops as a first-line treatment option. For these cases, a treatment algorithm involving gradual tapering of steroid drops and the consideration of steroid-sparing agents such as cyclosporine, lifitegrast, and tacrolimus has been suggested.
Since its debut, use of this drug has continued to climb due to its clinical success and increasing list of indications. Many severe cases of atopic dermatitis otherwise recalcitrant to treatment have shown significant skin improvement with dupilumab. Therefore, these patients are often reluctant to discontinue this life-changing therapy. In most cases, management of the ocular surface complications can allow a patient to continue their systemic treatment.
In most cases, the management of ocular surface complications has allowed patients to continue their systemic treatment with Dupixent. Despite the challenges, it is crucial for eye care providers to be aware of the possible ocular findings associated with Dupixent use and to actively manage and treat these ocular complications to enable patients to continue their therapy effectively.
Treatment: dupixent before and after photos
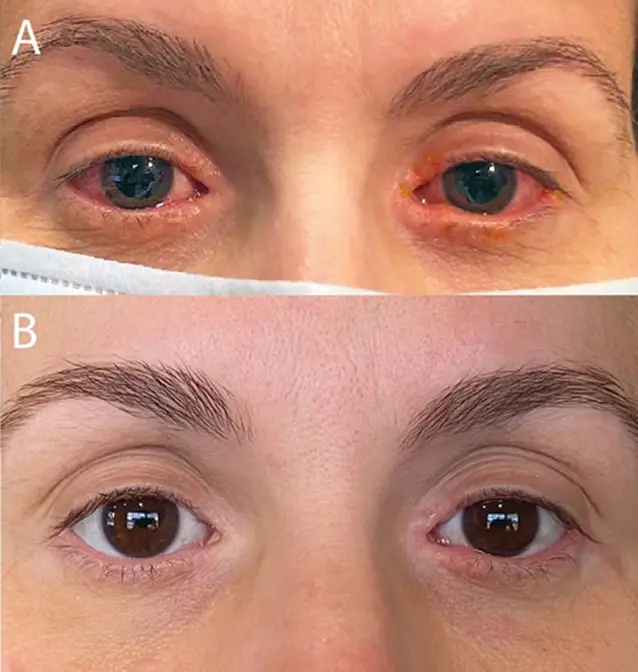
Fig. 1. (A) Initial clinic visit reveals bilateral conjunctival injection, epiphora and blepharitis. (B) Patient-provided photograph with significant improvement three months after initiation of dedicated ophthalmic therapy (image credit)
Try Eye Disease Diagnosis Quiz ; Follow us in Facebook
To read more
Discover more from An Eye Care Blog
Subscribe to get the latest posts sent to your email.


You must be logged in to post a comment.